Testing Plants as Potential Medicines
Background: In nature, organisms are constantly battling for resources and survival. Plants compete with other plants for light and water. Fast growth, big leaves, and large root systems would be advantageous characteristics. Some plants, such as rhododendrons, actually produce toxic chemicals that drip from their leaves into the soil, killing competing plants around them.
All organisms are infected by viruses or threatened by bacterial disease. Most organisms have defense systems to protect themselves from potential predators or foreign invaders. Plants such as fungi and bacteria produce antimicrobial agents to battle the microbes. If humans could find and isolate an antimicrobial molecule, it could potentially lead to a potential therapeutic medicine.
Once plant samples are collected, extraction techniques have to be determined. Then, samples can be tested for their ability to kill different types of microbes. Researchers need to prove that certain antimicrobial agents do not cause toxic effects in humans.
To test plant extracts for antimicrobial properties, technicians add extract-soaked filter paper disks to bacteria cultures spread on Petri plates. The extracts demonstrating these clear areas on the Petri dishes are then further purified and screened for the specific ingredients causing the bacterial death. These compounds may include antiseptics, astringents, antibiotics, and toxins.
Purpose: What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials:
-Plant extract (lemon tree leaves)
-syringe
-filter paper
-Petri dishes
-funnel
-microfuge tube
-rack
-sterilizer/autoclave
-filter paper disks
-pipet
-Bunsen burner and gas lighter
-glass spreader
-methanol
-E coli PROCEDURE:
Preparing Plant Extracts:
-Using a mortar and pestle, grind up 2 grams of plant tissue with 10 ml of deionized water
-Let it sit for 3 minutes
-Filter the sample through an 11 cm filter paper/funnel.
-Filter sterilize the extract using a syringe filter.
-Collect 1 ml of the filter-sterilized extract into a 1.7 microtube. Label the sample.
Preparing Plant Extract
Filter Sterilization Steps:
-Attach pre-filter to syringe and rinse with water
Take to Laminar Hood:
-Plant extract, syringe/prefilter, microfuge tube, rack, pipet
-Label microfuge tube (W&M)
-Attach sterile filter to the pre-filter
-Load 1.7 mls of extract into syringe, using pipet
-Depress plunger... greater than or equal to 1.0 ml of filter sterilization extract
- Snap on cap on microfuge tube.
-Evaporate methanol from methanol extracts by placing tube, with cap open, on a 65 Degrees C heat block overnight.
-Reconstitute methanol extract with 1.0ml sterile deionized water.
-Using sterile forceps, place 3 sterile pieces of filter paper in each extract tube.
-Store tubes at 4 Degrees C until ready to use.
Preparing Agar Plates
-Draw a “T” on each plate bottom and number the quadrants
-Liquefy sterile LB agar in the microwave.
-Using sterile technique, pour approximately 20ml of agar into Petri Plate.
-Label agar plates with a cross, label quadrants.
-Using sterile forceps, add the appropriate # of sterile disks to each tube of filtered extract.
-Prepare negative control disks:
2-sterile water
2- amphicillin
-Place the disks into the appropriate solution.
-Using sterile forceps, sterile disks were added to the microfuge tubes containing one ml sterile water and 1 ml ampicillin.
- 10-20ml of warmed nutrient agar was poured into 2 petri dishes using sterile technique.
- After allowing agar to solidify, plates were turned upside down and stored at 4 Degrees C overnight.
-1 ml of E coli colony was added to each plate. A flame-sterilized spreading loop was used to spread the bacteria throughout the surface of the agar.
-Using flame-sterilized forceps, filter disks were placed in separate quadrants onto the plate in the following sequence: 1) water
2) plant extracts
3) ampicillin
Plants were left on lab bench for 2 minutes to allow both bacteria and filter disks to adhere to the agar.
-Plants were inabated upside down, over night at 37 degrees C. Plates were photographed and observed for clearance around the filtered disks after 24 hours, 48 hours, and 72 hours.
Results:
Day 1 (24 hours):
Background: In nature, organisms are constantly battling for resources and survival. Plants compete with other plants for light and water. Fast growth, big leaves, and large root systems would be advantageous characteristics. Some plants, such as rhododendrons, actually produce toxic chemicals that drip from their leaves into the soil, killing competing plants around them.
All organisms are infected by viruses or threatened by bacterial disease. Most organisms have defense systems to protect themselves from potential predators or foreign invaders. Plants such as fungi and bacteria produce antimicrobial agents to battle the microbes. If humans could find and isolate an antimicrobial molecule, it could potentially lead to a potential therapeutic medicine.
Once plant samples are collected, extraction techniques have to be determined. Then, samples can be tested for their ability to kill different types of microbes. Researchers need to prove that certain antimicrobial agents do not cause toxic effects in humans.
To test plant extracts for antimicrobial properties, technicians add extract-soaked filter paper disks to bacteria cultures spread on Petri plates. The extracts demonstrating these clear areas on the Petri dishes are then further purified and screened for the specific ingredients causing the bacterial death. These compounds may include antiseptics, astringents, antibiotics, and toxins.
Purpose: What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials:
-Plant extract (lemon tree leaves)
-syringe
-filter paper
-Petri dishes
-funnel
-microfuge tube
-rack
-sterilizer/autoclave
-filter paper disks
-pipet
-Bunsen burner and gas lighter
-glass spreader
-methanol
-E coli PROCEDURE:
Preparing Plant Extracts:
-Using a mortar and pestle, grind up 2 grams of plant tissue with 10 ml of deionized water
-Let it sit for 3 minutes
-Filter the sample through an 11 cm filter paper/funnel.
-Filter sterilize the extract using a syringe filter.
-Collect 1 ml of the filter-sterilized extract into a 1.7 microtube. Label the sample.
Preparing Plant Extract
Filter Sterilization Steps:
-Attach pre-filter to syringe and rinse with water
Take to Laminar Hood:
-Plant extract, syringe/prefilter, microfuge tube, rack, pipet
-Label microfuge tube (W&M)
-Attach sterile filter to the pre-filter
-Load 1.7 mls of extract into syringe, using pipet
-Depress plunger... greater than or equal to 1.0 ml of filter sterilization extract
- Snap on cap on microfuge tube.
-Evaporate methanol from methanol extracts by placing tube, with cap open, on a 65 Degrees C heat block overnight.
-Reconstitute methanol extract with 1.0ml sterile deionized water.
-Using sterile forceps, place 3 sterile pieces of filter paper in each extract tube.
-Store tubes at 4 Degrees C until ready to use.
Preparing Agar Plates
-Draw a “T” on each plate bottom and number the quadrants
-Liquefy sterile LB agar in the microwave.
-Using sterile technique, pour approximately 20ml of agar into Petri Plate.
-Label agar plates with a cross, label quadrants.
-Using sterile forceps, add the appropriate # of sterile disks to each tube of filtered extract.
-Prepare negative control disks:
2-sterile water
2- amphicillin
-Place the disks into the appropriate solution.
-Using sterile forceps, sterile disks were added to the microfuge tubes containing one ml sterile water and 1 ml ampicillin.
- 10-20ml of warmed nutrient agar was poured into 2 petri dishes using sterile technique.
- After allowing agar to solidify, plates were turned upside down and stored at 4 Degrees C overnight.
-1 ml of E coli colony was added to each plate. A flame-sterilized spreading loop was used to spread the bacteria throughout the surface of the agar.
-Using flame-sterilized forceps, filter disks were placed in separate quadrants onto the plate in the following sequence: 1) water
2) plant extracts
3) ampicillin
Plants were left on lab bench for 2 minutes to allow both bacteria and filter disks to adhere to the agar.
-Plants were inabated upside down, over night at 37 degrees C. Plates were photographed and observed for clearance around the filtered disks after 24 hours, 48 hours, and 72 hours.
Results:
Day 1 (24 hours):
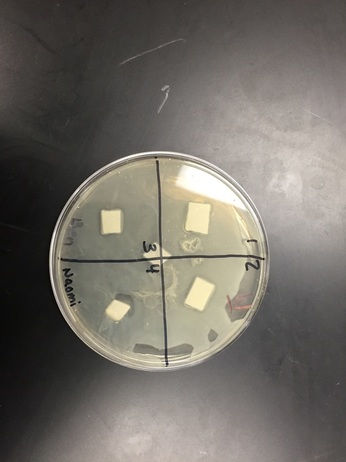
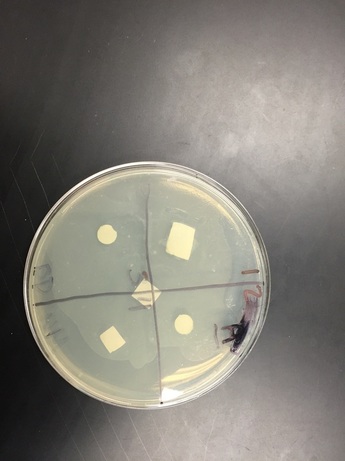
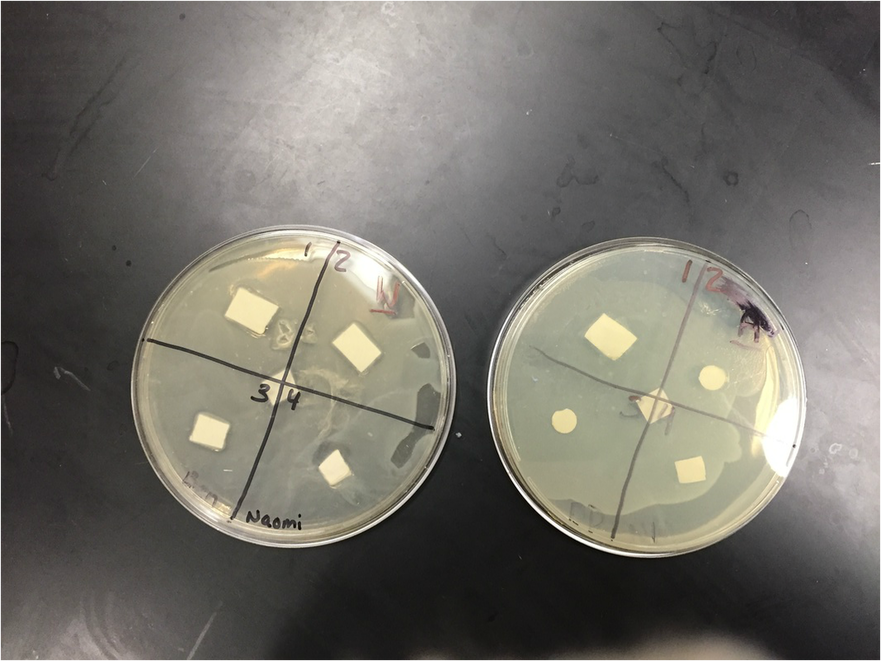
Agar: There was no clearance in any of the quadrants. There weren't any visible changes. The bacteria didn't spread that well either.
Water: There was a good amount of clearance in quadrant four. The bacteria seemed to have spread pretty evenly.
Water: There was a good amount of clearance in quadrant four. The bacteria seemed to have spread pretty evenly.
Water: There was lots of clearance is quadrant four. Some clearance was appearing in the other 3 quadrants, but there wasn't a lot.
Agar: Still no clearance. Bacteria hasn't spread all the way. The bacterial lawn did not grow completely and was spotty, making it hard to see if there was clearance.
Analysis/Conclusions:
With our water petri dish there was a clearing around the ampicillin. Ampicillin is a antimicrobial compound that causes death of bacterium E. coli by inhibiting the cell wall synthesis. The bacterial lawn didn't grow right up to the negative control disk. (That's where the clearance was located.) Water would not be expected to have antimicrobial activity, which makes it a good negative control for this experiment. I didn't see any clearings around the disks soaked in plant extract. There was only a clearing around the ampicillin. A possible problem that might have affected the results is that something wasn't sterile. I thought that this lab would have much more effective and enjoyable if we would have done it faster. This lab took way too long and the process became tedious. If we were to do this experiment again, we should get it done in 1 or 2 days, so that we are being more productive. If we get it done faster there will be a higher likelihood that the experiment will be successful. Even though one of my petri dishes had some clearance, I wasn't very impressed with the results because we didn't really go over what the results meant in class.
Agar: Still no clearance. Bacteria hasn't spread all the way. The bacterial lawn did not grow completely and was spotty, making it hard to see if there was clearance.
Analysis/Conclusions:
With our water petri dish there was a clearing around the ampicillin. Ampicillin is a antimicrobial compound that causes death of bacterium E. coli by inhibiting the cell wall synthesis. The bacterial lawn didn't grow right up to the negative control disk. (That's where the clearance was located.) Water would not be expected to have antimicrobial activity, which makes it a good negative control for this experiment. I didn't see any clearings around the disks soaked in plant extract. There was only a clearing around the ampicillin. A possible problem that might have affected the results is that something wasn't sterile. I thought that this lab would have much more effective and enjoyable if we would have done it faster. This lab took way too long and the process became tedious. If we were to do this experiment again, we should get it done in 1 or 2 days, so that we are being more productive. If we get it done faster there will be a higher likelihood that the experiment will be successful. Even though one of my petri dishes had some clearance, I wasn't very impressed with the results because we didn't really go over what the results meant in class.